Hydrophobic Interaction Chromatography
Hydrophobic Interaction Chromatography
Hydrophobic interaction chromatography (HIC) is a chromatography method according to differences in proteins surface hydrophobicity by utilizing a reversible interaction between these proteins and the hydrophobic surface of a HIC medium.
Main characteristics
-
Easy to use in combination with other chromatography methods to purify complex biological samples
-
Low sample pretreatment requirements, suitable for the early stages of the entire purification protocol
-
High dynamic binding capacity, low non-specific adsorption
Factors affecting HIC
1. Solid phase
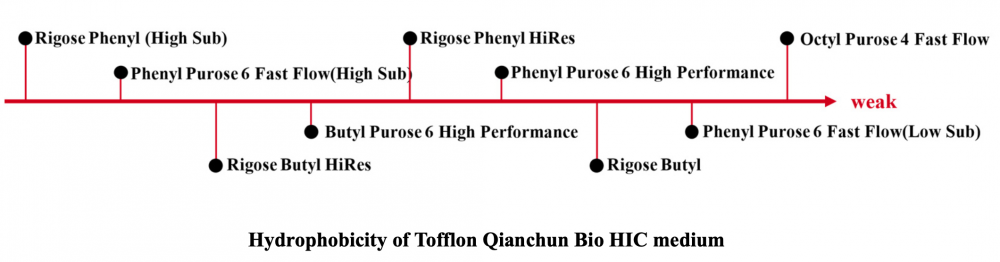
The selection of HIC chromatography medium is mainly based on the hydrophobic strength of the target molecule, and the chromatography medium with strong hydrophobicity is selected when separating the molecules with weak hydrophobicity, and the chromatography medium with weak hydrophobicity is selected when separating the molecules with strong hydrophobicity.
The hydrophobicity of common functional groups in hydrophobic chromatography media is: Phenyl > Octyl > Butyl > Butyl-S. In addition to the type of functional group, the type of matrix and the degree of substitution of functional group will also affect the sample separation effect.
2. Mobile phase
The addition of “salting-out” salts to the sample promotes ligand- protein interactions in HIC. As the concentration of salt is increased, the amount of bound protein increases up to the precipitation point for the protein. The most commonly used salts are (NH4)2SO4, CHCOONH4, Na2SO4 and NaCl. At a given concentration, ammonium sulfate often gives the best resolution of a mixture of standard proteins compared to other salts. Elution of bound solutes can be achieved simply by stepwise or gradient elution with buffers which have low salt content. Low recovery can be remedied by adding some organic modifiers in the elution buffers, such as 30% isopropanol.

The Hofmeister series of some anions and cations arranged according to their effects on the solubility of protein in aqueous solutions.
3. Purification condition
In addition to the solid phase and the mobile phase, other chromatographic conditions such as temperature and flow velocity will also have a certain effect on the chromatography. Usually with the increasing of temperature, the hydrophobic interaction will enhance. However, too high temperature will affect the conformation and solubility of the protein, so it is recommended to maintain a constant temperature during the separation and purification process. HIC is less sensitive to the flow velocity, so the flow velocity is mainly selected according to the separation time and the degree of medium tolerance.